Why Medications Affect People Differently: The Real Reasons Behind Drug Side Effects

Drug Metabolism Checker
Check Your Drug Metabolism Risk
This tool estimates your risk of experiencing adverse drug reactions based on common genetic variations that affect how your body processes medications. Results are based on published pharmacogenomic guidelines.
Key Information:
Have you ever taken the same medication as someone else - maybe a friend or family member - and had completely different experiences? One person feels fine, while the other ends up in the hospital. It’s not just bad luck. It’s biology. And it’s more common than you think.
Why Your Body Reacts Differently to the Same Drug
Two people take the same pill. Same dose. Same condition. But one gets sick, and the other doesn’t. Why? Because drugs don’t work the same way in every body. The difference isn’t about willpower, lifestyle, or even how well you follow instructions. It’s rooted in your genes, your age, your other medications, and even your recent illness.
When a drug enters your system, your body has to process it. That’s called pharmacokinetics - what your body does to the drug. Then, the drug interacts with its target - maybe a receptor in your brain, heart, or liver. That’s pharmacodynamics - what the drug does to your body. Both steps vary wildly between individuals.
Genetics play a huge role. Studies show genetic factors explain anywhere from 20% to 95% of how people respond to specific drugs. For example, the CYP2D6 enzyme is responsible for breaking down over 25% of commonly prescribed medications, including antidepressants, beta-blockers, and codeine. About 5-10% of people of European descent are "poor metabolizers" - their bodies barely break down these drugs, leading to dangerous buildup. On the flip side, 1-2% of Europeans and up to 29% of Ethiopians are "ultra-rapid metabolizers." Their bodies clear the drug so fast that it never reaches effective levels. For someone on codeine, that means no pain relief. For someone else, it can mean a fatal overdose.
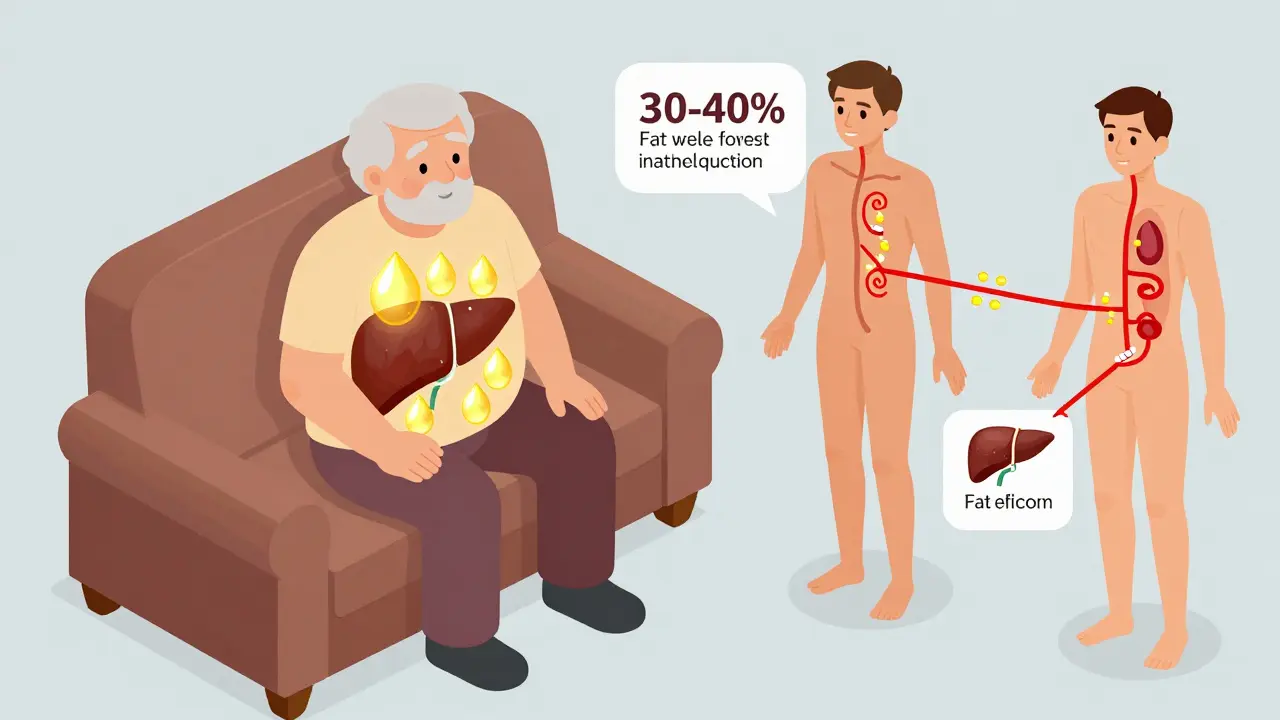
Age, Body Fat, and How Your Body Holds Onto Drugs
Age isn’t just a number when it comes to medication. As you get older, your body changes. Muscle mass drops. Fat increases. Liver and kidney function slow down. These shifts change how drugs are absorbed, distributed, and removed.
Older adults have 30-40% more body fat than younger people. That means fat-soluble drugs - like some anti-anxiety medications or cholesterol-lowering statins - get trapped in fatty tissue. The result? The drug sticks around longer, building up over time. That’s why seniors are more likely to experience dizziness, confusion, or falls from medications that seem perfectly safe for younger people.
And it’s not just age. If you’re overweight, underweight, or have chronic kidney or liver disease, your drug levels can be off - even if your doctor prescribed the "standard" dose.
What You’re Taking (and What You’ve Been Sick With)
It’s not just your genes and age. What else you’re taking matters. A drug interaction can turn a safe medication into a danger.
Take warfarin, a blood thinner. It’s tricky because small changes in its level can cause bleeding or clots. One study found a 68-year-old woman kept having dangerous spikes in her INR (a measure of blood clotting) - even though her dose hadn’t changed. Genetic testing revealed she had two copies of a rare variant in the CYP2C9 enzyme gene. That meant her body processed warfarin 60% slower than average. Once her dose was cut, her INR stabilized. No more hospital visits.
But genetics aren’t the whole story. Inflammation from an infection - even a simple cold - can reduce CYP enzyme activity by 20-50%. That means your body suddenly becomes a poor metabolizer overnight. A drug that was fine last week might now build up to toxic levels.
And polypharmacy? Taking five or more medications? That’s a recipe for disaster. Elderly patients on multiple drugs have a 300% higher risk of serious side effects. Why? Because each drug can interfere with how others are processed. Amiodarone, a heart medication, can block the metabolism of warfarin, doubling or even tripling its concentration. That’s not rare. It’s predictable - if you know what to look for.
The Rise of Pharmacogenomics: Personalized Medicine Is Here
Pharmacogenomics is the science of using your genes to predict how you’ll respond to drugs. It’s not science fiction. It’s already being used - and saving lives.
For example, clopidogrel (Plavix) is used after heart attacks to prevent clots. But 2-15% of people carry a genetic variant that makes the drug useless. Their bodies can’t activate it. Without testing, they’re left vulnerable to another heart attack - thinking the drug is working. Testing for the CYP2C19 variant can prevent that. The FDA has approved point-of-care tests that give results in under an hour.
And it’s not just heart drugs. In pediatric cancer, testing for TPMT and NUDT15 variants before giving mercaptopurine (used for leukemia) has cut severe toxicity from 25% to 12%. That’s a 52% drop in life-threatening side effects.
The FDA now includes pharmacogenomic information in the labels of over 300 drugs. For 44 of them, there are specific dosing guidelines based on genetics. Warfarin, clopidogrel, statins, antidepressants, and certain painkillers all have genetic recommendations.
Why Isn’t Everyone Getting Tested?
If this works so well, why aren’t we doing it for everyone?
Because the system isn’t ready.
Only 18% of U.S. insurers cover pharmacogenomic testing. Many doctors haven’t been trained to interpret the results. A 2023 survey found 68% of physicians felt unprepared to use genetic data in prescribing. Even when results are available, electronic health records often don’t flag risky interactions.
And here’s the hard truth: testing just three genes (CYP2C9, CYP2C19, CYP2D6) explains only 15-19% of all adverse drug reactions. There are thousands of genes involved. A single-gene test gives you a piece of the puzzle - not the whole picture.
That’s why experts are moving toward polygenic risk scores. Instead of looking at one gene, they combine hundreds - even thousands - of tiny genetic signals to predict how you’ll respond. Early studies show these scores improve prediction by 40-60% over single-gene tests. But they’re still in development. And expensive.

The Real Cost of Not Knowing
It’s not just about health. It’s about money.
In the UK, adverse drug reactions cost the NHS £770 million a year. In the U.S., they’re the fourth leading cause of death. A 2022 Mayo Clinic study of 10,000 patients found those who got genetic testing had 32% fewer emergency room visits and 26% shorter hospital stays.
And it’s not just hospital bills. Think about asthma. About 15% of severe asthma patients have a genetic variant in the LTC4 synthase gene. For them, leukotriene modifiers like zafirlukast can improve lung function by 45%. But for the other 85%? No benefit. Yet these drugs cost $250-$300 a month. That’s wasted money. And wasted time.
When we treat everyone the same, we’re not just guessing - we’re risking lives.
What’s Next? The Future Is Polygenic
The field is moving fast. In 2024, Medicare in the U.S. started covering pharmacogenomic testing for 17 high-risk medications. The European Union now requires pharmacogenomic data in all new clinical trials. The NIH’s Pharmacogenomic Resource for Clinical Care has data from over 1.2 million patients.
Costs have dropped too. A full pharmacogenomic panel that cost $2,000 in 2015 now runs around $250. As prices keep falling, testing will become routine - not optional.
But the biggest shift isn’t just in testing. It’s in mindset. We’re moving away from "one-size-fits-all" prescribing. We’re learning that your genes, your age, your other meds, and even your last cold all shape how a drug affects you. The future isn’t about finding the "best" drug. It’s about finding the right drug - for you.
Why do some people get side effects from drugs while others don’t?
It’s because of differences in genetics, age, body composition, other medications, and even recent illnesses. For example, some people have gene variants that make them poor or ultra-rapid metabolizers of drugs, meaning their bodies process medications too slowly or too quickly. Others may have liver or kidney issues that change how drugs are cleared. Even a simple infection can temporarily reduce how fast your liver breaks down a drug, leading to unexpected side effects.
Is genetic testing for drug reactions worth it?
For some people, yes - especially if you’re taking drugs like warfarin, clopidogrel, certain antidepressants, or chemotherapy agents. Studies show genetic testing can prevent up to 30% of serious side effects. For example, testing before starting clopidogrel can prevent heart attacks in people who don’t respond to it. Testing for warfarin sensitivity can cut dangerous bleeding events by over 30%. The cost of testing is now under $250, and for high-risk patients, it can save thousands in hospital bills.
Can I get tested for how I’ll react to medications?
Yes, but it’s not yet routine. Some hospitals and clinics offer pharmacogenomic testing, especially in cardiology, oncology, and psychiatry. You can also get tested through private labs - though insurance coverage is still limited. Talk to your doctor if you’ve had bad side effects before, are on multiple medications, or have a family history of drug reactions. A pharmacist or genetic counselor can help you decide if testing makes sense for you.
Do older adults react differently to medications?
Yes. As we age, our bodies change: we have more body fat, less muscle, slower liver and kidney function, and often take multiple drugs. These changes mean medications stay in the body longer and can build up to toxic levels. That’s why seniors are more likely to experience dizziness, falls, confusion, or bleeding from drugs that are fine for younger people. Doses often need to be lowered - not because they’re weak, but because their bodies process drugs differently.
Are there drugs that are especially risky because of genetics?
Yes. Warfarin (blood thinner), clopidogrel (antiplatelet), codeine (painkiller), and certain antidepressants like SSRIs are known to have strong genetic links. For example, people with two copies of the CYP2C9*3 variant need 60% less warfarin. Those with CYP2D6 ultra-rapid metabolism can overdose on codeine. People with HLA-B*57:01 are at high risk of a deadly skin reaction from abacavir (an HIV drug). Testing for these variants is recommended before prescribing.
Why isn’t everyone tested before getting a prescription?
Because the healthcare system isn’t set up for it. Many doctors aren’t trained to interpret genetic results. Electronic health records rarely flag risks. Insurance often won’t pay for testing. And while testing for a few genes helps, it doesn’t explain all side effects - there are thousands of genes involved. Until we have better tools, broader coverage, and better integration into daily care, testing remains selective - not standard.





Comments (12)
Stephanie Paluch
12 Mar 2026
I had no idea my body could turn a simple painkiller into a nightmare 😳 My grandma took codeine for back pain and ended up in the ER. Turns out she’s an ultra-rapid metabolizer. We never connected the dots until now. This post literally saved my life. 🙏
tynece roberts
13 Mar 2026
so like i was on like 5 meds last year and kept getting dizzy as hell and my doc just said "youre old" but like... what if its not age? what if its like my liver saying nope? i think we need to stop blaming symptoms on age and start testing. also i misspelled like 3 words but u get the point lmao
Hugh Breen
13 Mar 2026
This is the single most important medical conversation we’re NOT having. 🇬🇧 I work in NHS pharmacy and see this daily. A 72-year-old woman on statins + amiodarone + warfarin? She’s a walking time bomb. We’re treating patients like they’re widgets on an assembly line. Pharmacogenomics isn’t futuristic - it’s basic hygiene. Let’s stop pretending one-size-fits-all works. We’re literally killing people with laziness.
Byron Boror
13 Mar 2026
This is why America’s healthcare is better. We don’t need to test every damn gene. People just need to take responsibility. If you’re allergic to a drug, don’t take it. If you’re old, take less. Simple. Stop overcomplicating everything with science. We don’t need DNA reports to know not to mix alcohol with benzos.
Lorna Brown
15 Mar 2026
It’s not just genetics. It’s the entire system. We’ve turned medicine into a transactional factory. Doctors have 7 minutes per patient. No time to ask about your last cold, your supplements, your cousin’s weird reaction to the same pill. We’re reducing human biology to checkboxes. And then we’re shocked when people die. This isn’t about technology - it’s about humility. We don’t know everything. And pretending we do is dangerous.
Rex Regum
15 Mar 2026
Oh wow, so now we’re going to DNA-test everyone before they get a Tylenol? Next thing you know, they’ll scan your iris before you buy aspirin. This is how socialism creeps in. "We know better than your body." Bullshit. My body’s been fine for 40 years on the same meds. Stop pathologizing normal people. You’re creating panic where there’s none.
Kelsey Vonk
15 Mar 2026
I’ve been on antidepressants for 12 years. Tried 7 different ones. One gave me panic attacks. One made me sleep 18 hours. One just… did nothing. I never knew why. This post made me cry. Not because I’m sad - but because I finally understand. It wasn’t me being broken. It was biology. And that’s okay. I’m not broken. My genes are just… different. 🌱
Emma Nicolls
17 Mar 2026
omg i just remembered my mom took that one heart med and got super sick and they thought it was her diet but turns out she had that gene variant? and they just kept giving her more? i think we need to tell more people about this. also i love how this post just explains everything without being like super academic. so refreshing
Jimmy V
18 Mar 2026
Let’s cut the fluff. Testing CYP2D6/CYP2C9/CYP2C19 costs $150. Saves $20k in ER visits. ROI is 130x. If your insurance won’t cover it, demand it. If your doctor doesn’t know about it, fire them. This isn’t "personalized medicine" - it’s basic safety. We test babies for phenylketonuria. Why not test adults before giving them life-altering drugs? Stop being lazy. Your life matters.
Richard Harris
19 Mar 2026
I’ve been on warfarin for 8 years. My INR was wild until my pharmacist suggested testing. Turned out I’m a slow metabolizer. Dose halved. No more bleeding. I didn’t even know testing existed. I’m just glad I found it. Thanks for sharing this - it’s not common knowledge, but it should be.
Kandace Bennett
21 Mar 2026
Honestly? This is why I love the U.S. We’re leading the world in precision medicine. Other countries are still stuck in the 1980s. Meanwhile, we’ve got FDA-approved point-of-care tests, Medicare coverage, and real data from 1.2 million patients. If you’re not getting tested, you’re not being proactive. And if you’re against it? You’re choosing ignorance. 🇺🇸💎
Tim Schulz
22 Mar 2026
Ah yes, the classic "let’s test everyone’s DNA before they take a pill" - because nothing says progress like turning your doctor’s office into a sci-fi lab. Meanwhile, in the real world, people are still getting prescribed antibiotics for colds. But sure, let’s spend $250 to find out why you got dizzy after a headache pill. I’ll wait for the hologram prescription.