Author: Bamberger Tynan - Page 2
Learn how to identify authorized generics by checking packaging, labeling, and NDC codes. Understand why they're identical to brand-name drugs but sold under different labels.
MedWatch and VAERS are two separate FDA systems for tracking side effects - one for drugs and devices, the other for vaccines. Learn how they work, why they're kept apart, and how to use them responsibly.
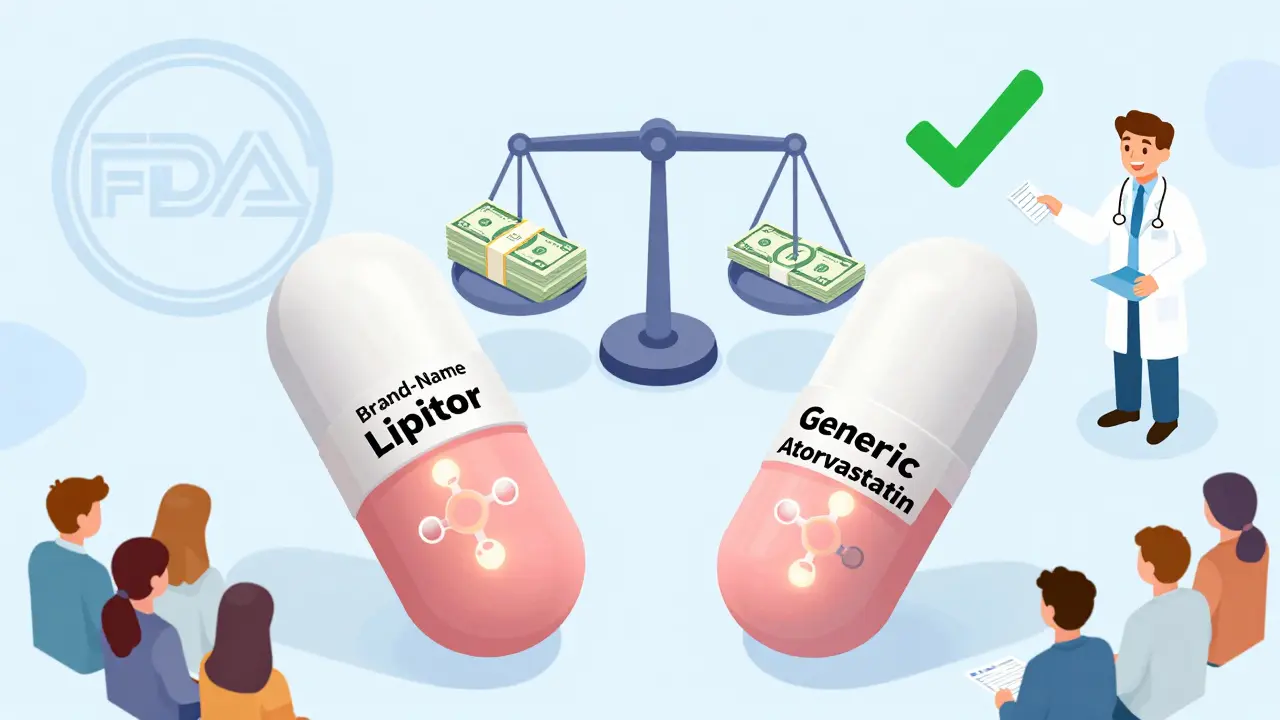
Generic drugs are just as effective as brand-name medications for most people, meeting the same strict FDA standards for safety and performance. They save billions annually and are used in 90% of U.S. prescriptions.
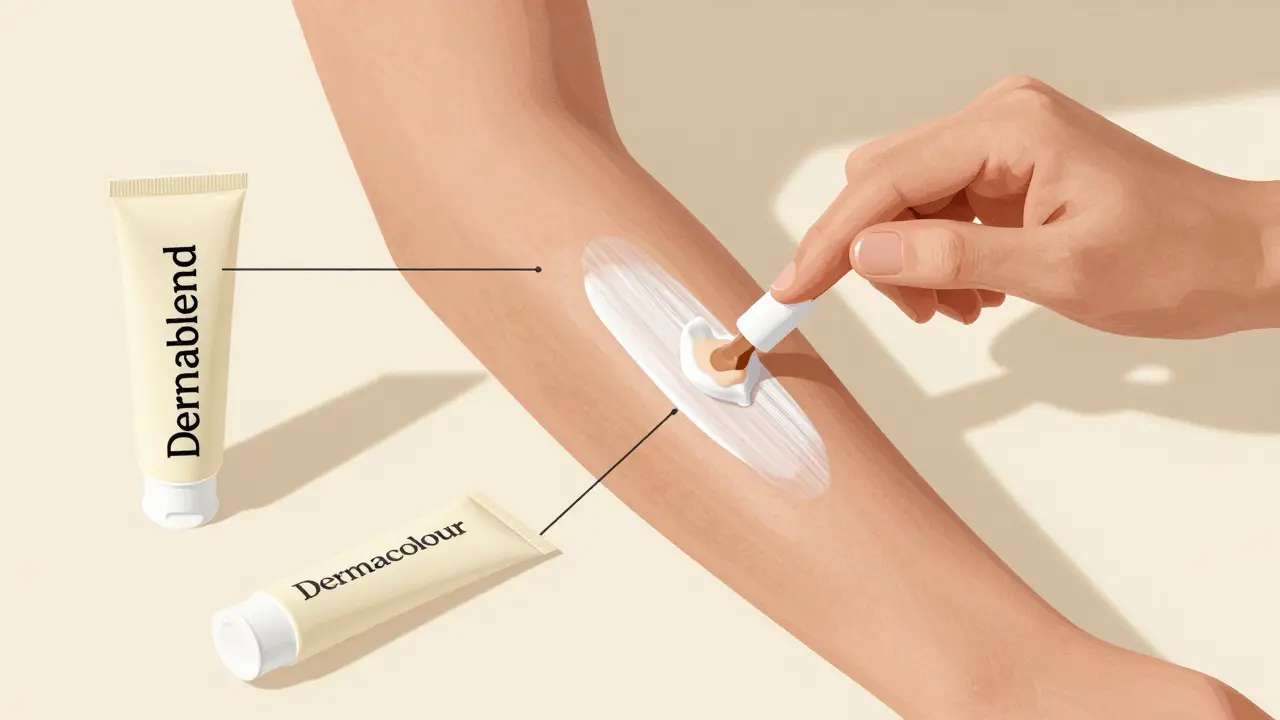
Vitiligo camouflage uses specialized cosmetics and micropigmentation to blend depigmented patches with surrounding skin. Learn which products work, how to apply them, and whether permanent tattooing is right for you.
Eustachian tube dysfunction causes ear pressure, muffled hearing, and popping. Learn how to relieve symptoms naturally, when to see a doctor, and what treatments actually work - from chewing gum to balloon dilation.
Targeted and cellular therapies are transforming leukemia and lymphoma care, offering deeper remissions and even cures where chemotherapy failed. Learn how these breakthroughs work-and who benefits most.
Learn how to securely store high-risk medications like opioids and benzodiazepines to prevent accidental overdoses in your home. Simple steps like using locked boxes, keeping pills in original containers, and tracking doses can save lives.
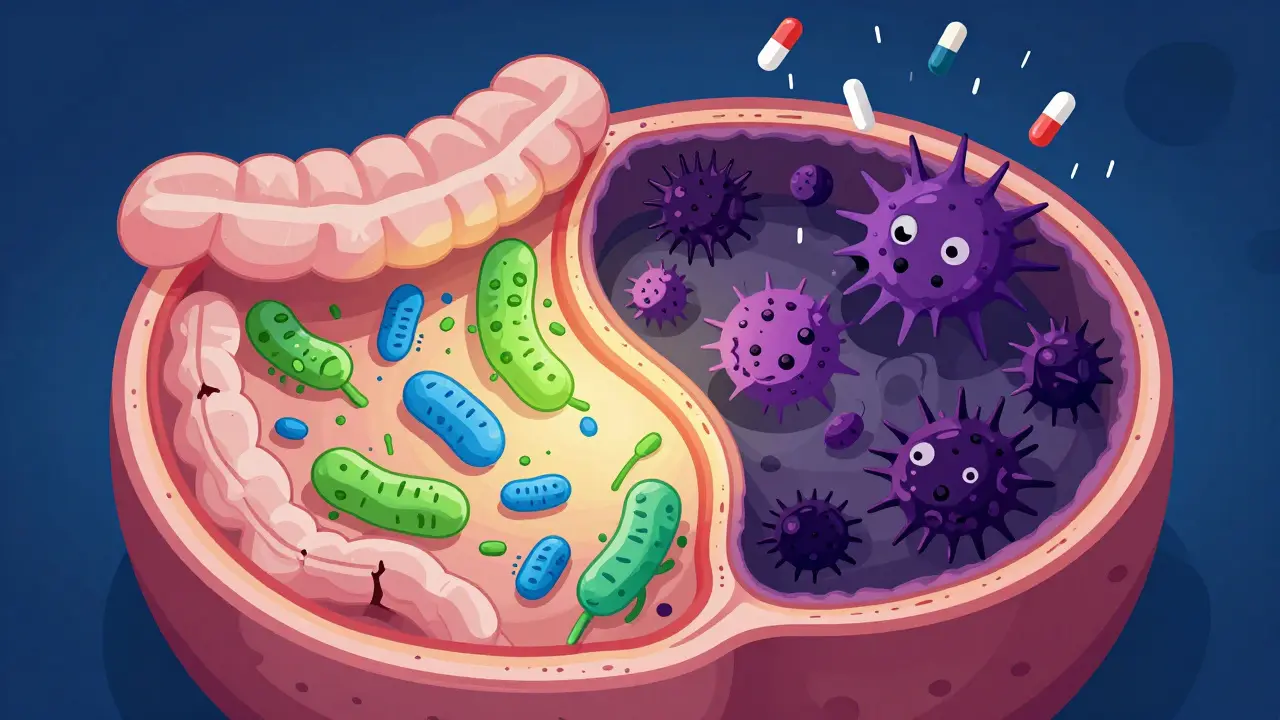
C. difficile colitis is a dangerous infection often triggered by antibiotics. Learn which drugs carry the highest risk and how fecal transplants offer a 90% cure rate for recurrent cases.
Idiopathic hypersomnia is a rare neurological disorder causing extreme daytime sleepiness despite long sleep. Learn about its symptoms, how it differs from narcolepsy, current treatments like Xywav, and why diagnosis often takes years.
The FDA Orange Book lists approved generic drugs and tells pharmacists which ones can be safely swapped for brand-name drugs. It's the key to affordable medication in the U.S.
Heat and humidity can degrade medications long before their expiration date, reducing potency and risking your health. Learn which drugs are most vulnerable and how to store them safely.
Support groups and community programs significantly improve medication adherence by offering peer-driven emotional and practical support. Research shows they outperform traditional education methods, reduce hospitalizations, and cut healthcare costs - especially when culturally matched and led by trained peers.