Category: Pharmacy - Page 2
Learn how to save up to 92% on generic azithromycin. Compare prices from GoodRx, RedBox Rx, and Canadian pharmacies to find the cheapest safe options online.
Learn how drug-drug interactions happen through pharmacokinetic and pharmacodynamic mechanisms, the role of CYP450 enzymes, and how to prevent dangerous medication clashes.
Explore how authorized generics impact pharmaceutical competition, disrupt 180-day exclusivity, and influence patent litigation settlements.
Learn how to identify authorized generics by checking packaging, labeling, and NDC codes. Understand why they're identical to brand-name drugs but sold under different labels.
The FDA Orange Book lists approved generic drugs and tells pharmacists which ones can be safely swapped for brand-name drugs. It's the key to affordable medication in the U.S.
Digital tools now help prevent dangerous interactions between generic drugs, OTC meds, and supplements. Learn how Epocrates, Micromedex, and DDInter work - and why they’re changing patient safety.
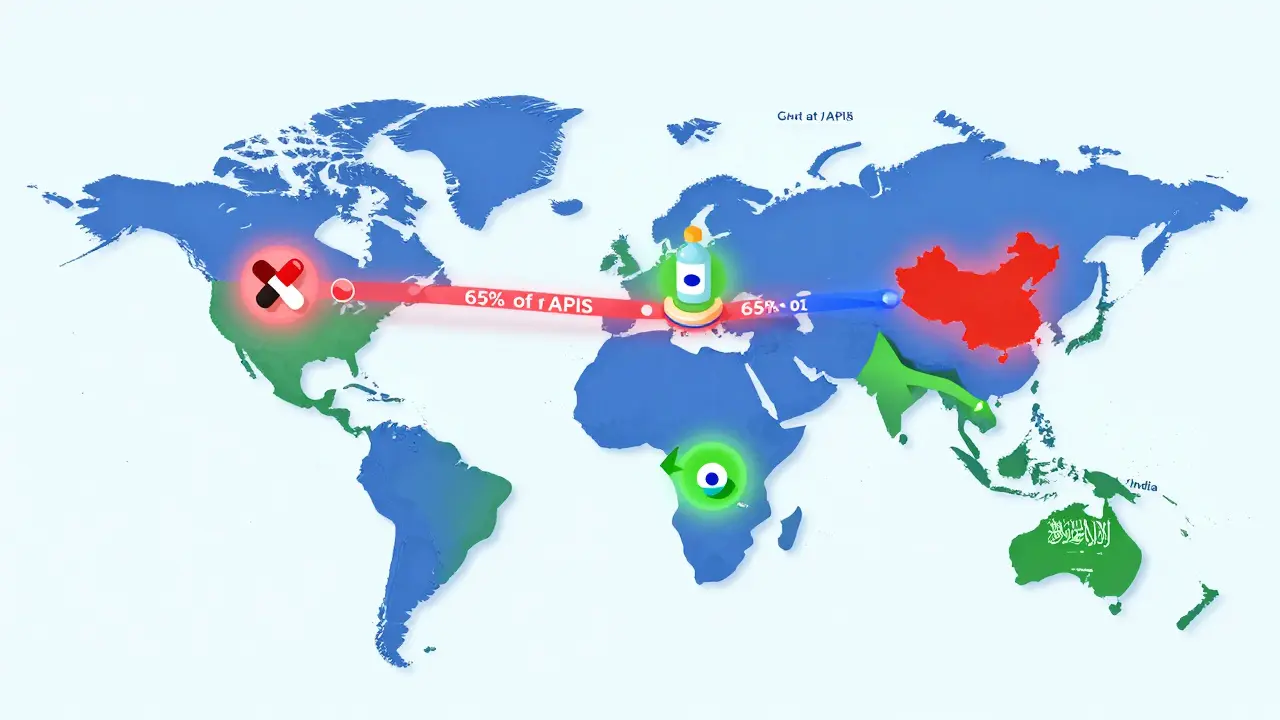
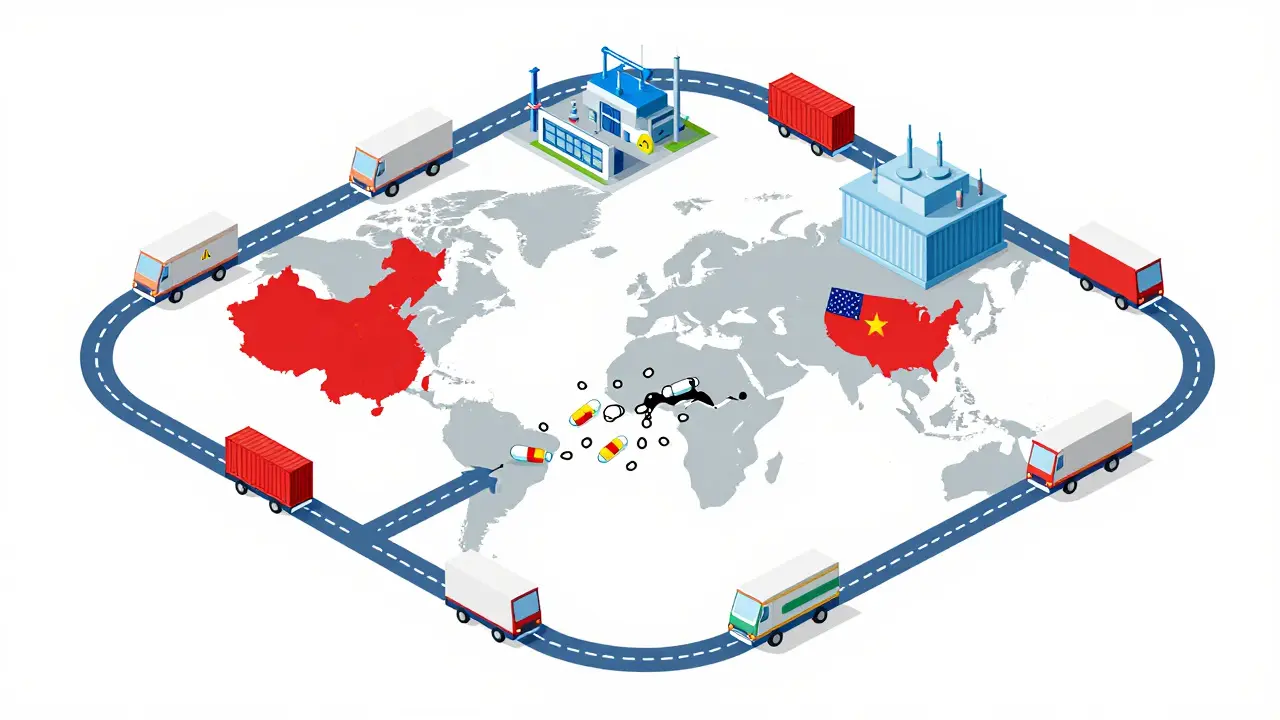
The global generic drugs market is evolving fast. With biosimilars rising, supply chains shifting, and emerging economies driving growth, affordability remains key - but quality and innovation are now just as important.
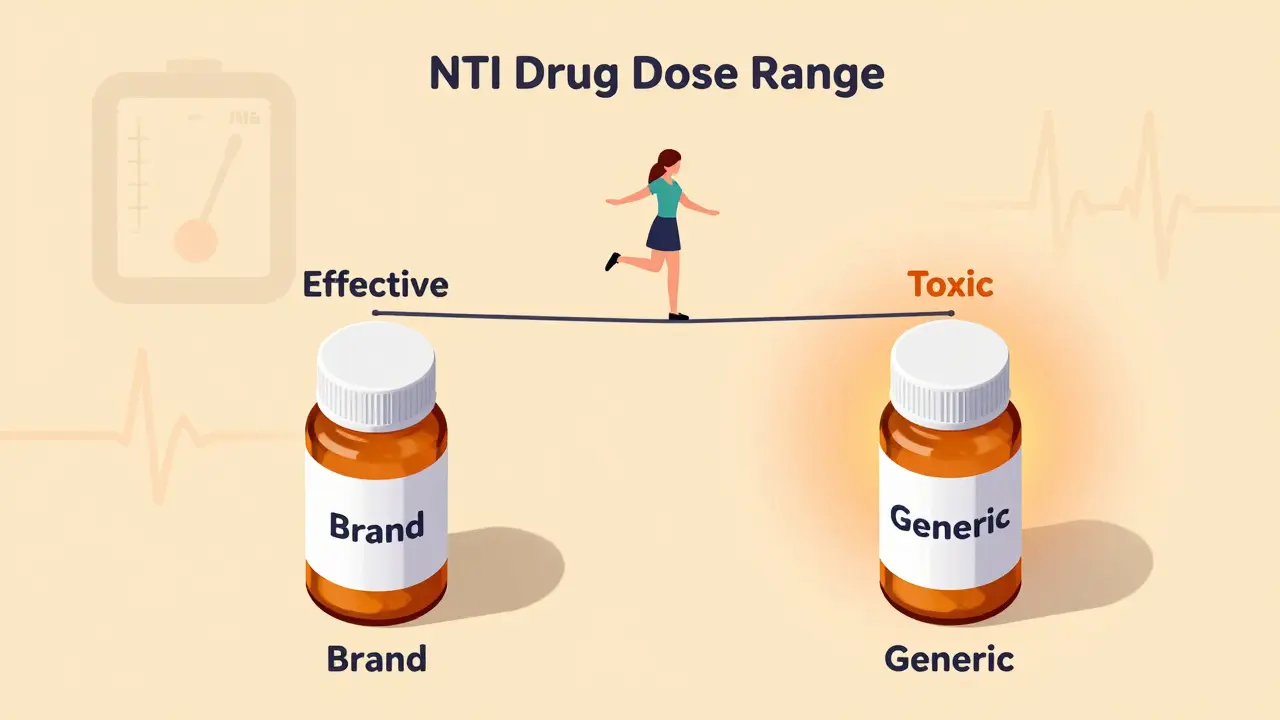
NTI drugs like warfarin and phenytoin have a tiny margin between effective and toxic doses. Generic substitutions can push patients out of this narrow range, risking serious harm - even death. Here’s why switching isn’t always safe.
Pharmacists can legally substitute generic drugs in all U.S. states, but therapeutic substitution rules vary widely. Learn how state laws shape what pharmacists can do, the documentation required, and why this matters for patient access and safety.
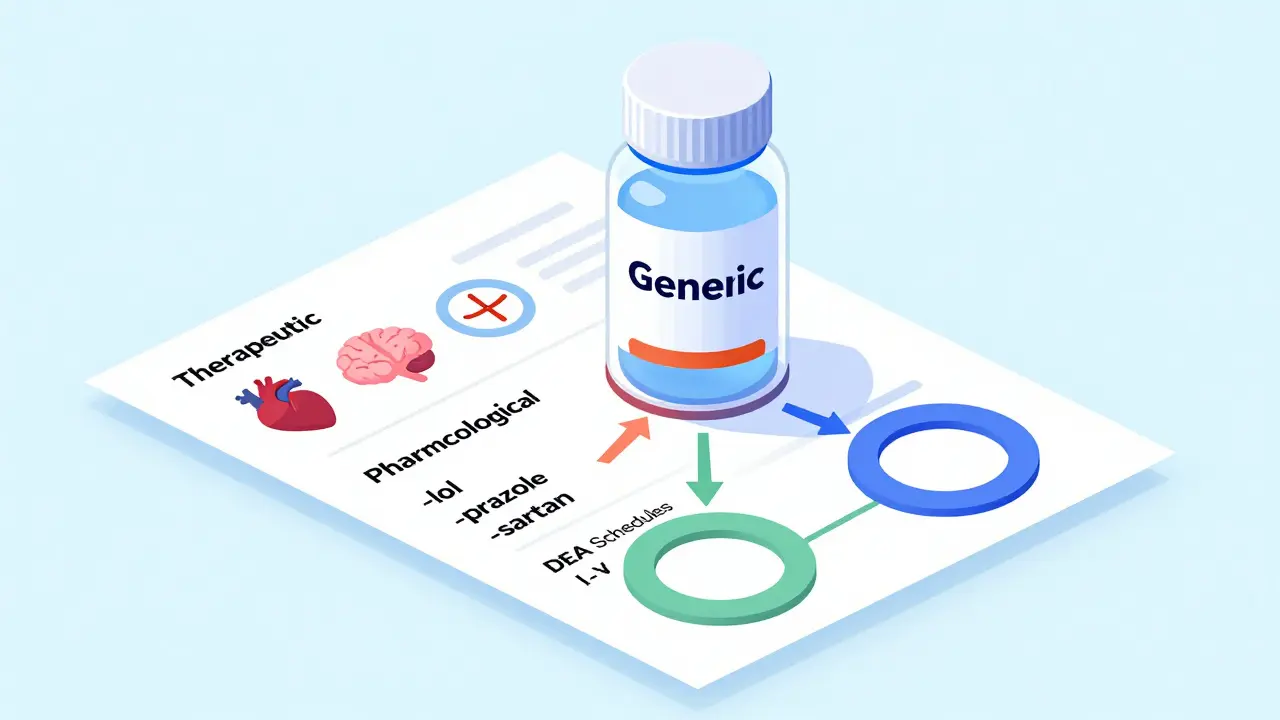
Generic drugs are classified by therapeutic use, mechanism of action, legal status, and insurance tiers. Learn how these systems work, why they matter, and how they affect your prescriptions.
Europe's tendering system for generic drugs prioritizes transparency, fairness, and value over lowest price. Learn how MEAT evaluation, framework agreements, and digital tools shape public pharmaceutical procurement across the EU.
Learn how to prevent drug shortages by building resilient pharmaceutical supply chains through buffer stocks, dual-sourcing, AI forecasting, and regional manufacturing. Real strategies for companies and policymakers.